PhD proposal
PhD Director: Pierre Joseph – LAAS-CNRS
PhD co-supervisor: Paolo Bergese – Università degli Studi di Brescia, Italy
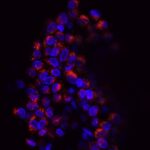
Extracellular Vesicles (EVs) are nanoparticles released by cells, key players in intercellular communication and in the maintenance of tissue homeostasis and in pathogenesis1. EVs contribute to cancer initiation and progression: they modulate cancer-associated processes (immunosuppression, angiogenesis, invasion, and metastasis). Hence, EVs and their biomolecular cargo can be detected in body fluids and exploited as biomarkers in cancer.
However, little is known about EV genesis by tumors. We make the educated guess that EV inception may depend on high compressive stress that can be found in cancer, such as pancreatic adenocarcinoma (PDAC), a disease with no cure. They may reveal as very promising class of non-invasive biomarkers, for early diagnosis.
We also hypothesize that, regardless of their cargo, EV structure and size distribution can be considered as a distant looking glass into physical constraints within hard-to-reach tumors such as PDAC. This offers therapeutic opportunities for patient management, especially as in PDAC, mechanical stress increases resistance to treatment.
Collectively, this interdisciplinary thesis aims at characterizing the role of physical constraints in EV production in experimental models of pancreatic cancer, up to patient cohorts.
Objectives:
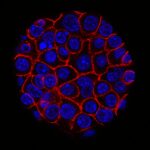
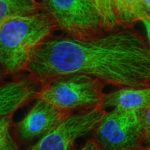
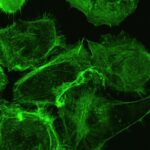
- Generate tumoroids (tumor spheroids) of human and mouse-derived pancreatic cancer cells using routine techniques.
- Analyze the influence of different stresses (mechanical pressure, flow, chemicals), on the size and distribution of EV produced by tumoroids, thanks to microfluidic chips.
- Detect and analyze EVs within real-life cohorts, first in blood from mice with PDAC, then in blood from patients with full clinical annotation and ancillary resources.
Key words: cancer, extracellular vesicles, microfluidic